|
Cancer Cell Adhesion and Mechanics
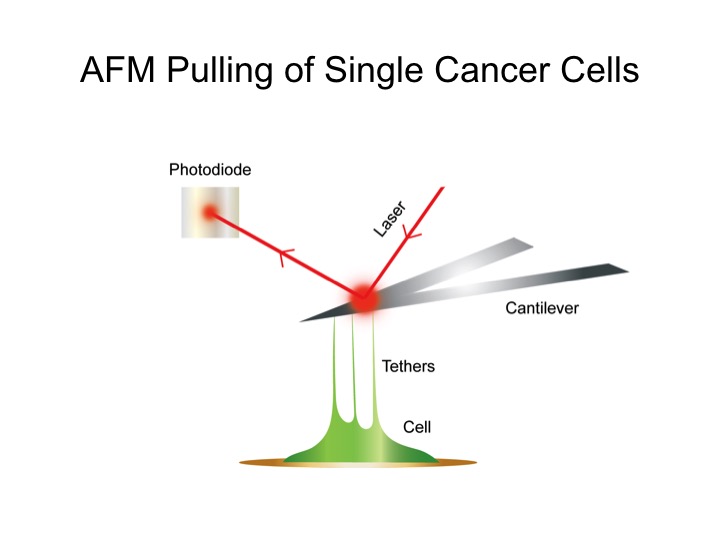
Cancer microenvironment plays a critical role in the last step of cancer metastasis – colonization. Recreating the cancer microenvironment mimicking the conditions for in vitro studies of cancer metastasis would provide valuable knowledge on how cancers spread. Cell adhesion force is a key parameter for cancer attachment and, therefore, may be used as the potential diagnostic and prognostic marker for cancer metastasis. We are interested in how physical parameters, such as cell elasticity and viscosity, change with different stages of cancer. We use single cell force measurement technique to quantify the effect of the mechanics of the environment on cancer cell adhesion. Overall, this research may ultimately provide pertinent information on the key steps in cancer metastasis in a more physiological relevant context, thus improving upon current approaches in therapeutic development. The project will improve our understanding of the effect of cancer microenvironment on cancer invasion and metastasis. The knowledge gained should allow us to further establish basis for building platforms mimicking in vivo conditions for in vitro studies of cancer metastasis, biomarker discoveries and cancer drug screening.
Projects
Protein Mechanical Activation
 Von Willebrand factor (VWF) is a large glycoprotein that is responsible for blood clotting and thrombosis. To maintain hemostasis, VWF mediates platelet adhesion to the subendothelial connective tissue that lines the interior of all blood vessels and binds to the clotting factor VIII. As a glycoprotein, it can reach up to 100 micrometers in length and can exhibit tremendous flexibility in its structure. VWF function is activated by an allosteric mechanism or by high-shear induced force. Beyond the allosteric model, VWF are generally regulated by hydrodynamic forces within the blood vessels. In blood vessels, the shear rate of fluid is at its maximum near the subendothelial wall. Shear-induced adhesion enables VWF to bind to the endothelial under pathophysiological conditions and to initiate the blood clotting process at the site of vessel damage. We are studying VWF and ultra-large VWF resistance to mechanical forces and the effect of shear induced property changes. We will also study the VWF properties in the presence of ADAMTS-13 to test its reductase activity.
DNA Melting and Phase Transition
The DNA-gold nanoparticle system is a model for phase transitions. Melting of short, free DNA is not a phase transition. However, when short DNA are bound to gold particles, the system undergoes a phase transition because the DNA-gold particles form networks of micrometer size, which is approaching a bulk phase. Thus, the binding transition in the network is much sharper than that of free DNA in solution, due to the cooperative melting process. Many fundamental aspects of phase transitions may be investigated with this biomolecular system.
|

